In this link: http://rua.ua.es/dspace/handle/10045/51725, it is possible to acces to a very easy to use Graphical User Interface (GMcal_TieLinesLL v.2.3) based on the topological information contained in the Gibbs energy of mixing function, that has been developed for binary and ternary LLE systems, as a friendly tool to check the coherence of the parameters obtained in a correlation data procedure (the NRTL and UNIQUAC models are included by defect, however any other GExcess(L)/RT model could be easily implemented).
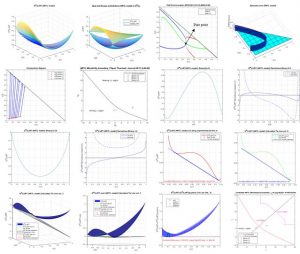
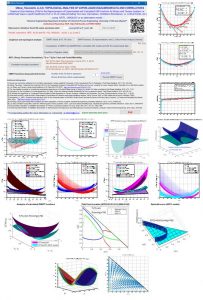
The analysis of the GM(L)/RT surface in the whole range of composition, the GM(L)/RT for the binaries subsystems and the GM(L)/RT curves in planes containing the liquid-liquid tie lines should be necessary to validate the obtained parameters for the different models for correlating phase equlibrium data. This simple analysis could be used by authors and journals in order to guarantee the adequate prediction of equilibrium. In addition, restrictions on NRTL binary parameters have been used in the correlation procedure to guarantee the adequate prediction of the total or partial miscibility behavior of the binary subsystems.
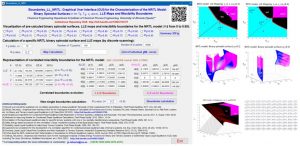
The NEW VERSION 2.3 of this friendly tool also allows the analysis of the Hessian Matrix determinant (σ), the spinodal curve (σ=0) and the Plait Point location for LLE ternary systems, using additionally the δ and δ* matrix determinants, which have to be also equal to zero and additionally the miscibility boundaries (for αij values different from 0.2). The analysis of LLE binary systems at different temperatures is also possible, including the location, if there exits, of the Critical Solution Temperature (UCST, LCST or Closed Misciblity Loops).
To follow updates and comments: LinkedIn Research Gate Facebook
** After using this GUI: GMcal_TieLinesLL, please complete the following quick inquiry to know your opinion about the graphical user interface developed: English version // Spanish version.
Related references:
Marcilla, A.; Labarta, J.A.; Olaya, M.M. Should we trust all the published LLE correlation parameters in phase equilibria? Necessity of their Assessment Prior to Publication. Fluid Phase Equilibria. 2017, 433, 243-252 (http://dx.doi.org/10.1016/j.fluid.2016.11.009). The 4th most cited article published in Fluid Phase Equilibria in 2017-2020 (extracted from Scopus 11/2020). Research Gate. RUA: http://hdl.handle.net/10045/66521.
Labarta, J.A.; Olaya, M.M.; Marcilla, A. What does the NRTL model look like? Determination of boundaries for different fluid phase equilibrium regions. AIChE Journal. 2022, 68(10), e17805 (https://dx.doi.org/10.1002/aic.17805).
Marcilla, A.; Labarta, J.A.; Serrano M.D.; Olaya, M.M. GE Models and Algorithms for Condensed Phase Equilibrium Data Regression in Ternary Systems: Limitations and Proposals. The Open Thermodynamics Journal. 2011, 5, (Suppl 1-M5) 48-62 (http://dx.doi.org/10.2174/1874396X01105010048).
Marcilla, A.; Serrano, M.D.; Labarta. J.A.; Olaya, M.M. Checking Liquid-Liquid Critical Point Conditions and their Application in Ternary Systems. Industrial & Engineering Chemistry Research. 2012, 51(13), 5098-5102 (http://dx.doi.org/10.1021/ie202793r).
Olaya, M.M.; Carbonell-Hermida, P.; Trives, M.; Labarta, J.A.; Marcilla, A. LLE data correlation using NRTL model for different types of binary systems: UCST, LCST and closed miscibility loops. Industrial & Engineering Chemistry Research. 2020, 59(17), 8469-8479 (https://doi.org/10.1021/acs.iecr.0c00141).
Vicente-Martínez, M.; Labarta, J.A. Análisis topológico del modelo NRTL en sistemas binarios y ternarios para caracterizar la tipología de los sistemas líquido-líquido que predice en función de los valores de los parámetros de interacción binaria. Institutional Repository of the University of Alicante (RUA) 2021. (http://hdl.handle.net/10045/117665).
Labarta, J.A.; Olaya, M.M.; Marcilla, A. Boundaries_LL_NRTL Graphical User Interface (GUI) for the characterization of the NRTL model: Binary Spinodal Surfaces (in the τi,j-τj,i-xi space), LLE maps and Miscibility Boundaries. Institutional Repository of the University of Alicante (RUA) 2022. (http://hdl.handle.net/10045/121471).
Labarta, J.A.; Olaya, M.M.; Marcilla, A. GMcal_TieLinesVL: Graphical User Interface (GUI) for the Topological Analysis of Experimental and Calculated GM Functions for Binary and Ternary (isobaric or isothermal) Vapor-Liquid Equilibrium (VLE) data (including Tie-Lines, Derivatives, Distillation Boundaries, LL Critical Points Location, etc.). Institutional Repository of the University of Alicante (RUA). 2022. Publicly available online at: http://hdl.handle.net/10045/122857.